Cancer research studies—including clinical trials—improve how we prevent, find, and treat cancer. They have brought us the treatments we have today. Here are the answers to some of the questions you may have about joining a research study or clinical trial.
What kinds of cancer research studies exist?
There are several kinds of cancer research studies, including clinical trials. Clinical trials test new ways to improve patient care. Some types of clinical trials include:
- Prevention Trials: Look at ways to reduce your chances of getting cancer.
- Screening Trials: Test ways to find cancer earlier, when it may be easiest to treat.
- Treatment Trials: Test new medicines or treatments or test new ways of using the medicines or treatments that already exist to find out: If they are safe, if they work as well as or better than what we now use, and what doses work best.
- Quality of Life or Supportive Care Trials: Test ways to improve the comfort and well-being of people living with cancer.
- Population Studies: Explore cancer patterns or trends.
Who can join a cancer research study?
There are many cancer research studies, and each one needs specific types of people to join. For example, a study might look for:
- People without cancer.
- People who may be at high risk of having cancer in the future.
- People who have just been told that they have cancer and have not yet started treatment.
- People who have finished standard treatment, but still have cancer and are looking for other new and possibly better treatment options to try.
- People who have family members with cancer.
Why is it important for cancer research studies to include people from all backgrounds?
We can all be touched by cancer.
How well medications and treatments work for someone can depend on things like their:
- Gender
- Race and ethnicity
- Age
- Weight
- Where they live
When research studies and clinical trials only include some groups of people, doctors will not know if medications or treatments are safe and if they will work for all groups of people. By having people from all backgrounds participate in cancer research studies, we can know more about how to best treat patients in the future. That is why it is so important for people of all backgrounds to be part of cancer research studies.
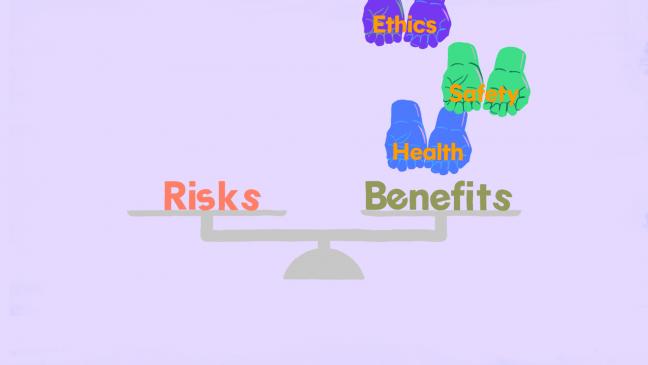
What are the potential benefits and risks of being in a cancer research study?
Being in a cancer research study can have both benefits and risks (or concerns).
- Benefits- In the study, you may get offered newer treatment or treatments that help people in new ways. You may also not get any direct benefit yourself from being in the study. For example, some people will get the new medicine or treatment being tested, while other people will not. Also, some treatments will not give any new benefit.
- Risks- Cancer researchers and doctors will tell you about any risks they know about before you join the study. This may include doing tests or treatments that are uncomfortable or inconvenient, as well as the chance that the treatment being tested is not any better than the treatment you would have received outside of the study. There may also be other risks that they don’t know about yet.
How safe is it to join a cancer research study?
All cancer research studies have safeguards in place to help protect the rights, welfare and safety of people who are in the study. One safeguard is that all research studies are carefully reviewed for safety, risks, and benefits by a group of researchers and community members.
Cancer researchers and doctors will also go over details of the research study—including details about safety and known risks—with you before you decide to join.
What will I know about a cancer research study before I decide to join?
Cancer researchers and doctors will tell you all about the research study before you join. When they do, they will review a consent form with you. The consent form will tell you:
- The purpose of the study.
- The known risks and side effects, as well as the safeguards to lessen these risks.
- The potential benefits.
- What to expect during the study, including information about any tests that will be done.
- How your personal information will be used and protected.
- That joining a research study is voluntary and that you can choose to stop at any time.
The cancer researchers and doctors will also take the time to answer any questions you may have before you sign the consent form. Keep in mind, you can always stop being part of a study.
How can I join a cancer research study?
Talk to your friends, family, and health care providers, then visit our current list of cancer research studies.
Sources: NIH.gov, Cancer.gov
Special thanks to the many Norris Cotton Cancer Center volunteers whose expertise facilitated the development of this webpage and video. This work is supported by the NCI, grant number 3P30CA023108-40S4