Borwell 338 West
Dartmouth Cancer Center
One Medical Center Drive
Lebanon, NH 03756
(603) 646-5503
Hours: Monday to Friday, 9 am to 5:30 pm
Authorized users have 24/7 access.
Pricing information and sample submission
Overview
The Dartmouth Cancer Center Microscopy Shared Resource (DCC MSR) provides access to advanced imaging instrumentation and image analysis software to Dartmouth Cancer Center members, and all members of Dartmouth College and Dartmouth Hitchcock Medical Center communities. The instrumentation available include point scanning confocal microscopy and Airyscan super resolution, automated multi-spectral image acquisition and spectral unmixing, high throughput phase contrast and widefield fluorescence imaging of live cells, conventional widefield fluorescence and brightfield imaging, as well as an UV light based micropatterning and microfabrication bioengineering system. Workstations for image analysis are available within the facility. To enable effective and safe use of the MSR resources, facility personnel are available for training of prospective users in image theory and hands-on use of specific instrumentation. Facility personnel are available for consultation on experimental design, data acquisition, and analysis as well as to perform data acquisition and analysis for interested labs.
Leadership
Radu V. Stan, MD, PhD
Director
Professor of Biochemistry and Cell Biology
Contact:
Rubin 623
Phone: (603) 646-5206
Fax: (603) 653-9952
Radu.V.Stan@Dartmouth.edu
Alexander S. George, PhD
Manager
Contact:
Borwell 338 West
(603) 646-5503
Alexander.S.George@Dartmouth.edu
Training
Facility staff provide an introductory imaging theory lecture set several times per year. Users who have completed this training may request hands-on training sessions to qualify for independent use. Please contact Alex George (Alexander.S.George@Dartmouth.edu) or Radu Stan (Radu.V.Stan@Dartmouth.edu) with a completed training request form (DOC) to schedule training.
Services
We provide consultation services for imaging experiments at no cost to users. Facility personnel are available to work with investigators using the microscopes at an additional hourly cost or to perform data acquisition and image analysis on a fee for service basis. Please contact Alex George (Alexander.S.George@Dartmouth.edu) or Radu Stan (Radu.V.Stan@Dartmouth.edu) to access these services.
Usage
Fully trained users may access microscope calendars and make reservations through RaDar and will be able to access the facility 24/7 via a card reader. Users in training may access the facility Monday to Friday, 9 am to 5:30 pm, by appointment with facility staff.
Important: Please acknowledge the Microscopy resource of the Dartmouth Cancer Center Irradiation, Imaging, Microscopy and Animal Cancer Models Shared Resource (RRID:SCR_023478) along with NCI Cancer Center Support Grant 5P30CA023108, in any publications or presentations. Users of the confocal microscope should also acknowledge the NIH shared instrumentation award 1S10OD021616.
Suggested text: “XXX was carried out in the Irradiation, Imaging, Microscopy and Animal Cancer Models Shared Resource (RRID:SCR_023478) at Dartmouth Cancer Center which is supported by NCI Cancer Center Support Grant 5P30CA023108 and NIH awards.”
Instrumentation
Zeiss LSM 800 confocal miscroscope with Airyscan
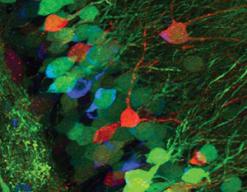
This microscope features classical confocal point scanning capability for optical sectioning and 3D reconstruction as well as sub-diffraction resolution capability through the Airyscan detector. Adjustable spectral filters and 4 lasers make this a suitable system for almost any dye in the visible range. An environmental chamber with temperature, humidity, and gas mixture control allows extended observation of live samples.
Akoya Vectra 3 automated multispectral pathology imaging system
This system is an upright automated slide scanner for scanning of up to six slides with chromogenic (i.e. H&E, IHC) or fluorescence staining. The instrumentation is capable of multi-spectral imaging and spectral unmixing, which dramatically decreases background and allows for a high degree of multiplexing of a broad range of fluorophores in the same sample. Vectra, Phenochart, inForm and HALO3 software is available for acquisition control and image analysis.
Cytation 5 automated imager with BioSpa8 incubator
This system houses up to 8 rectangular cell culture plates in a CO2 cell culture incubator and can image with brightfield, phase contrast, and up to four channels of florescence. Nitrogen is available to maintain hypoxic conditions for an additional cost. The system is ideal for performing high throughput long-term experiments on live samples without disrupting culture conditions.
Olympus IX83 / PRIMO micropatterning microfabrication
This microscope serves as a platform for a PRIMO bioengineering device. The PRIMO is capable of precise UV light-based patterning of molecules on any surface and microfabrication using hydrogels based over ranges from micron-scale features to centimeter-scale culture areas. An Andor Sona sCMOS camera allows alignment with sample features and monitoring of pattern progress.
Image analysis workstation
We offer an independent workstation for post-acquisition analysis featuring multiple image analysis packages licenses including HALO3, InForm and Gen5, as well as serving as a platform for open-source analysis workflows using ImageJ.